Type 1 diabetes mellitus (T1D) is an autoimmune disease, which is caused by the immune system mistakenly attacking pancreatic β cells, resulting in absolute insufficient insulin secretion, and patients need to rely on insulin injection for life. However, traditional treatment can only control blood glucose, which cannot reverse the process of β-cell damage and is difficult to prevent the continuous occurrence of autoimmune attack. In recent years, with the in-depth study of intestinal microecology, the key role of intestinal flora and its metabolites in the pathogenesis of T1D has been gradually revealed. In particular, the imbalance of intestinal microbiota-bile acid axis has been confirmed to be one of the core mechanisms driving the progression of the disease. In May 2025, Liu Yu’s team from Run Run Shaw Hospital Affiliated to Nanjing Medical University and Wu Hao’s team from Fudan University published a blockbuster study in Cell Reports Medicine, which brought a breakthrough dawn for the treatment of T1D by regulating bile acid metabolism. best blood glucose meter
1. The “gut derived” code for T1D: imbalance of the microbiota-bile acid axis
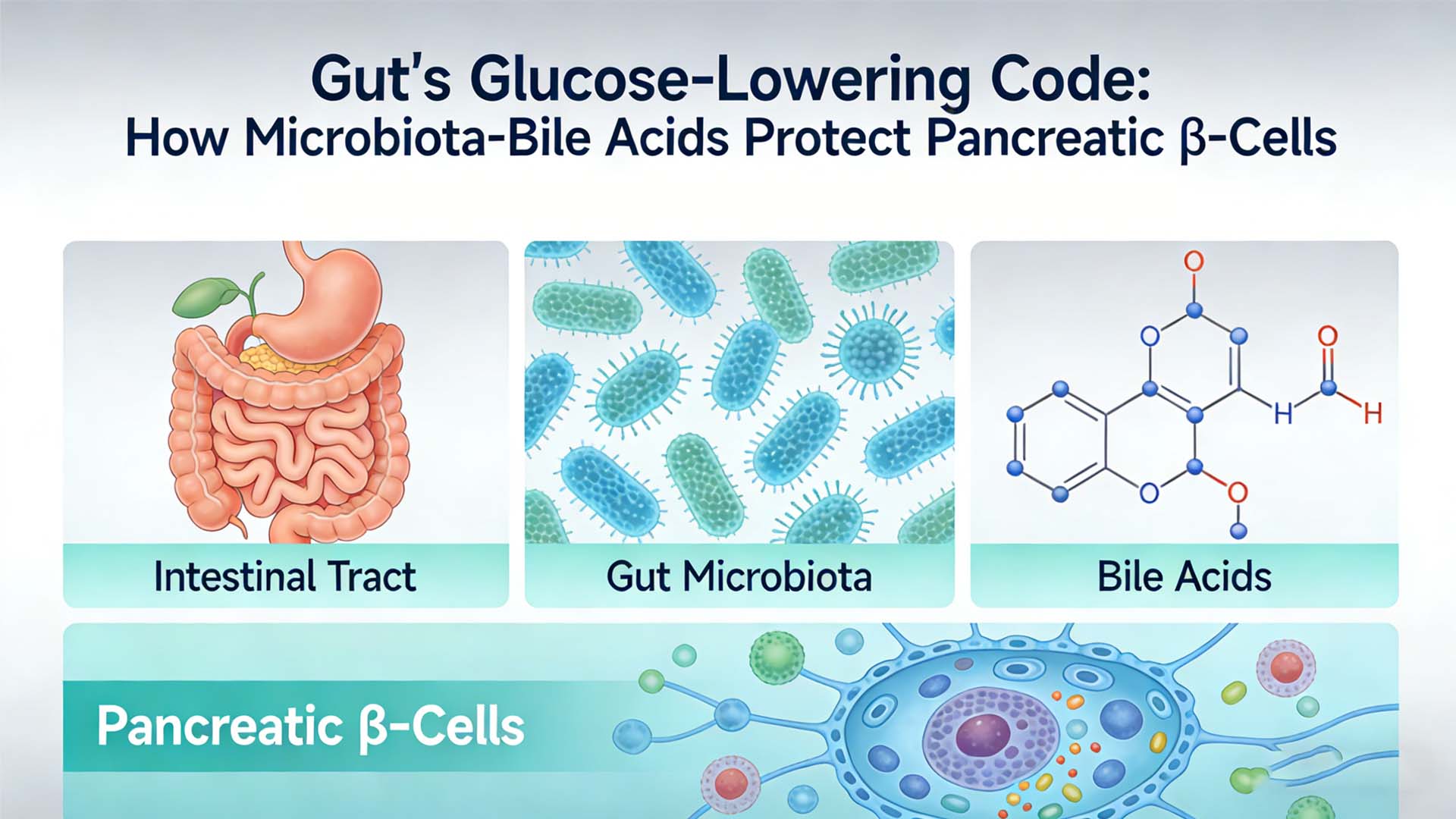
The gut is not only the core site of digestion and absorption, but also the largest immune organ and microecosystem of the human body. Intestinal microbiota is involved in the regulation of host immune function, glucose and lipid metabolism and islet β cell homeostasis by producing metabolites such as secondary bile acids and short-chain fatty acids.
Using metagenomic sequencing and targeted metabolomics analysis of 111 T1D patients and healthy controls, the team identified core abnormalities in the gut microbiome of T1D patients:
01 Dysbiosis of gut microbiota: Beneficial bacteria (Faecalibacterium prausnitzii, Roseburia faecis, etc.) are significantly reduced in the gut of T1D patients, while harmful bacteria such as Escherichia coli and Klebsiella pneumoniae are enriched, resulting in reduced gut microbial diversity and impaired barrier function.
02 Secondary bile acid metabolism disorders: Secondary bile acid synthesis pathways (such as bai operon, HSDH gene) are significantly down-regulated in the gut of T1D patients, resulting in the level of ** lithocholic acid (LCA), isolithocholic acid (isoLCA), deoxycholic acid (DCA) ** and other secondary bile acids being reduced by more than 50% compared with healthy people. Further analysis showed that the levels of these secondary bile acids were significantly positively correlated with the residual β-cell function (as reflected by C-peptide levels) in patients, suggesting that secondary bile acids are key regulatory molecules in maintaining islet function.
03 Potential for early diagnosis: The research team innovatively integrated five gut microbiota markers and three secondary bile acids to construct a “microbiota-bile acid” multi-omics prediction model. The AUC value of this model was 0.988, and it could accurately distinguish between T1D patients with preserved islet function (T1Dpos) and failure of islet function (T1Dneg), providing a new tool for early screening and accurate classification of the disease.
2. Microbiota transplantation: repair bile acid metabolism and reverse β-cell damage
Based on the above findings, the research team used ** washed fecal microbiota transplantation (WMT) ** technology to intervene in T1D patients, which is a rigorously screened and purified fecal microbiota transplantation technology, which can effectively avoid the safety risks of traditional fecal microbiota transplantation. blood glucose analyzer
Clinical results show that WMT therapy brings significant benefits to T1D patients:
01 Intestinal microecological reconstruction: After treatment, the abundance of beneficial bacteria such as Roseburia and Faecalibacterium in the gut of patients was significantly increased, the bile acid metabolic pathway returned to normal, and the levels of secondary bile acids (LCA and isoLCA) increased 2-3 times compared with those before treatment.
Improvement of islet function: C-peptide level was significantly increased in some patients, insulin dosage was reduced by 30% on average, blood glucose fluctuation was significantly reduced, and time in range (TIR) increased from 55% to 78%. Most importantly, the degree of improvement in islet function was significantly positively correlated with the increase in intestinal LCA and isoLCA levels, which directly confirmed the protective effect of secondary bile acids on β-cells.
Immunomodulatory effect: WMT treatment also reduced the levels of pro-inflammatory cytokines (IL-17A, IL-1β), reduced the proportion of Th17 cells, alleviated the autoimmune attack, and created a favorable environment for the repair of β cells.
3. Mechanism analysis: How do secondary bile acids protect islet β cells?
Secondary bile acids, as metabolites of gut microbiota, protect islet β cells through the following pathways:
01 can protect β cells directly. LCA and isoLCA can promote β cell proliferation, inhibit cell apoptosis and improve insulin secretion by activating G protein-coupled receptor 5 (TGR5).
02 Regulate immune balance: secondary bile acids can inhibit intestinal inflammation, reduce the release of pro-inflammatory factors, and regulate the function of dendritic cells and T cells, reduce the attack of autoimmune cells on β cells.
03 Improve intestinal barrier: secondary bile acids can enhance the expression of intestinal tight junction protein, repair intestinal barrier function, reduce bacterial endotoxin into the blood, thereby reducing systemic low-grade inflammation and improving insulin sensitivity.
4. Clinical significance and future prospects
This study is the first to systematically elucidate the dynamic regulation mechanism of the gut microbiota-bile acid axis in T1D, opening up new directions for the treatment of T1D:
01 New strategy of non-immune intervention: Different from traditional immunosuppressive therapy, it provides a safe and effective non-immune intervention program for T1D by regulating intestinal flora and bile acid metabolism, and avoiding the side effects caused by immunosuppression.
02 The possibility of precision treatment: Based on the microbiota-bile acid prediction model, early diagnosis and accurate classification of T1D can be achieved, and personalized treatment can be developed for patients.
Secondary bile acids and their receptors, such as TGR5, are expected to become new drug targets for the treatment of T1D. In the future, probiotics, prebiotics or small molecule drugs targeting bile acid metabolism can be developed to achieve precise regulation of T1D. blood glucose kit
The concept of treating glucose from the “intestine” breaks the limitations of traditional diabetes treatment, and brings new hope for T1D patients to reverse the disease. In the future, with the in-depth research on the mechanism of gut microbiota-bile acid-islet axis and the promotion of related clinical transformation, it is expected to achieve early diagnosis and treatment or even functional cure of T1D, so that more patients can get rid of lifelong dependence on insulin.
Post time: Feb-28-2026